Written by Steven Hansen
The U.S. new cases 7-day rolling average are 9.1 % HIGHER than the 7-day rolling average one week ago and U.S. deaths due to coronavirus are now 23.0 % LOWER than the rolling average one week ago. Today’s posts include:
- U.S. Coronavirus New Cases are 75,267
- U.S. Coronavirus deaths are at 932
- U.S. Coronavirus immunizations have been administered to 58.2 % of the population
- The 7-day rolling average rate of growth of the pandemic shows new cases improved and deaths improved
- US is making plans just in case the Covid-19 vaccines need a booster, officials say
- Pfizer CEO: Third dose of COVID-19 vaccine ‘likely’ needed within 1 year
- C.D.C. Panel Keeps Pause on Use of J&J Vaccine, Citing Need to Assess Potential Risks
- Doubtful COVID-19 vaccine makers can provide enough shots for all U.S. adults, despite their commitments to ship more than a billion doses
- US could have 300 million excess vaccine doses by the end of July
- Phase 3 trial finds corticosteroid inhaler speeds recovery time in COVID-19 patients
- Merck said Thursday that it has seen “encouraging” results in a clinical trial of an antiviral pill to treat Covid-19 early in the disease’s course
- Experts call for travel restrictions as COVID-19 cases surge in India
- Chloroquine is a potent inhibitor of SARS coronavirus infection and spread
- Vax Passports Are a Bad Idea
- IBM flags more cyber attacks on COVID vaccine infrastructure

include($_SERVER[‘DOCUMENT_ROOT’].’/pages/coronavirus1.htm’); ?>
Hospitalizations Are The Only Accurate Gauge
Hospitalizations historically appear to be little affected by weekends or holidays. The hospitalization growth rate trend is improving.

source: https://gis.cdc.gov/grasp/covidnet/COVID19_3.html
Historically, hospitalization growth follows new case growth by one to two weeks.
As an analyst, I use the rate of growth to determine the trend. But, the size of the pandemic is growing in terms of real numbers – and if the rate of growth does not become negative – the pandemic will overwhelm all resources.
The graph below shows the rate of growth relative to the growth a week earlier updated through today [note that negative numbers mean the rolling averages are LOWER than the rolling averages one week ago]. As one can see, the rate of growth for new cases peaked in early December 2020 for Thanksgiving, and early January 2021 for end-of-year holidays – and it now shows that the coronavirus effect is lower.

In the scheme of things, new cases decline first, followed by hospitalizations, and then deaths.
The New Variants Are The Primary Cause Of This Fourth Wave
Even with vaccinations picking up, the fourth wave is now underway.
- the more people that are vaccinated reduces the pool of people that can be infected. Today we have removed over 58 % of the population from being infected which theoretically should reduce the infection rate by 58 % [it is unproven whether the vaccines prevent a vaccinated person from being a carrier of the virus even though showing no signs]. If the vaccines are shown to stop transmission, then in theory it would reduce the infection rate by double the percent vaccinated [in this case you prevent your own infection and do not pass it along to another].
- it is also unknown what the effective rate of the current vaccines is against mutations that seem to appear almost daily. As an example, if the effective rate drops to 60%, it means the 58 % reduction in the infection rate discussed above is almost cut in half. The South African and Brazilian variant is reported somewhat immune to the current vaccines.
- In theory, the pandemic should be over immediately if everyone could be vaccinated today. The problem is that every day brings a new mutation (which would not appear if the pandemic was stopped). The longer the immunization process takes – the more ineffective the vaccine will become.
- It is not clear whether the vaccine prevents those vaccinated from spreading the virus. It seems to be well documented that it normally stops the virus from taking hold and when it does not – the infection is mild.
The real question is whether the vaccines will be mitigating this surge – and to what extent.
Coronavirus News You May Have Missed
C.D.C. Panel Keeps Pause on Use of J&J Vaccine, Citing Need to Assess Potential Risks – New York Times
The pause in the use of Johnson & Johnson’s Covid-19 vaccine may continue for a week to 10 days, after expert advisers to the Centers for Disease Control and Prevention determined on Wednesday that they needed more time to assess a possible link to a rare but serious blood-clotting disorder.
The decision not to reinstate the vaccine has painful consequences, nationally and globally. It may further erode public confidence in vaccination in general and slow the rollout of desperately needed shots to rural and underserved areas and homebound people. The vaccine is considered ideal for hard-to-reach people and places because it requires only one shot and is more easily stored and shipped than the vaccines made by Moderna and Pfizer-BioNTech, which must be kept at very low temperatures.
“Putting this vaccine on pause, for those of us that are frontline health care workers, has really been devastating,” Dr. Camille Kotton of Harvard Medical School told the panel. She said that losing the Johnson & Johnson vaccine even temporarily represented a big blow to efforts to stop the pandemic, especially in underserved communities.
… Advisory meetings usually end with a vote on whether or how to use a vaccine. But in this case, the panel members declined to vote after reviewing several options — including whether to limit use of the vaccine to older adults, as many European nations have done — saying that they did not have enough information to assess the potential risks.
… The panel of experts discussed the known background rates of low platelets and of the clots in the brain — known as central venous sinus thrombosis — in the general population and in younger women like those who had the clots, but noted that there was not enough data to precisely estimate how often the two conditions occurred at the same time.
… The safety bar for vaccines is set high because they are given to healthy people. The seemingly greater vulnerability of younger people to the clotting disorder is of particular concern, because their risk of severe illness from Covid itself is lower than that in older people. Those differences suggest that over all, compared to older people, younger people may have less to gain and more to lose from the Johnson & Johnson and AstraZeneca vaccines.
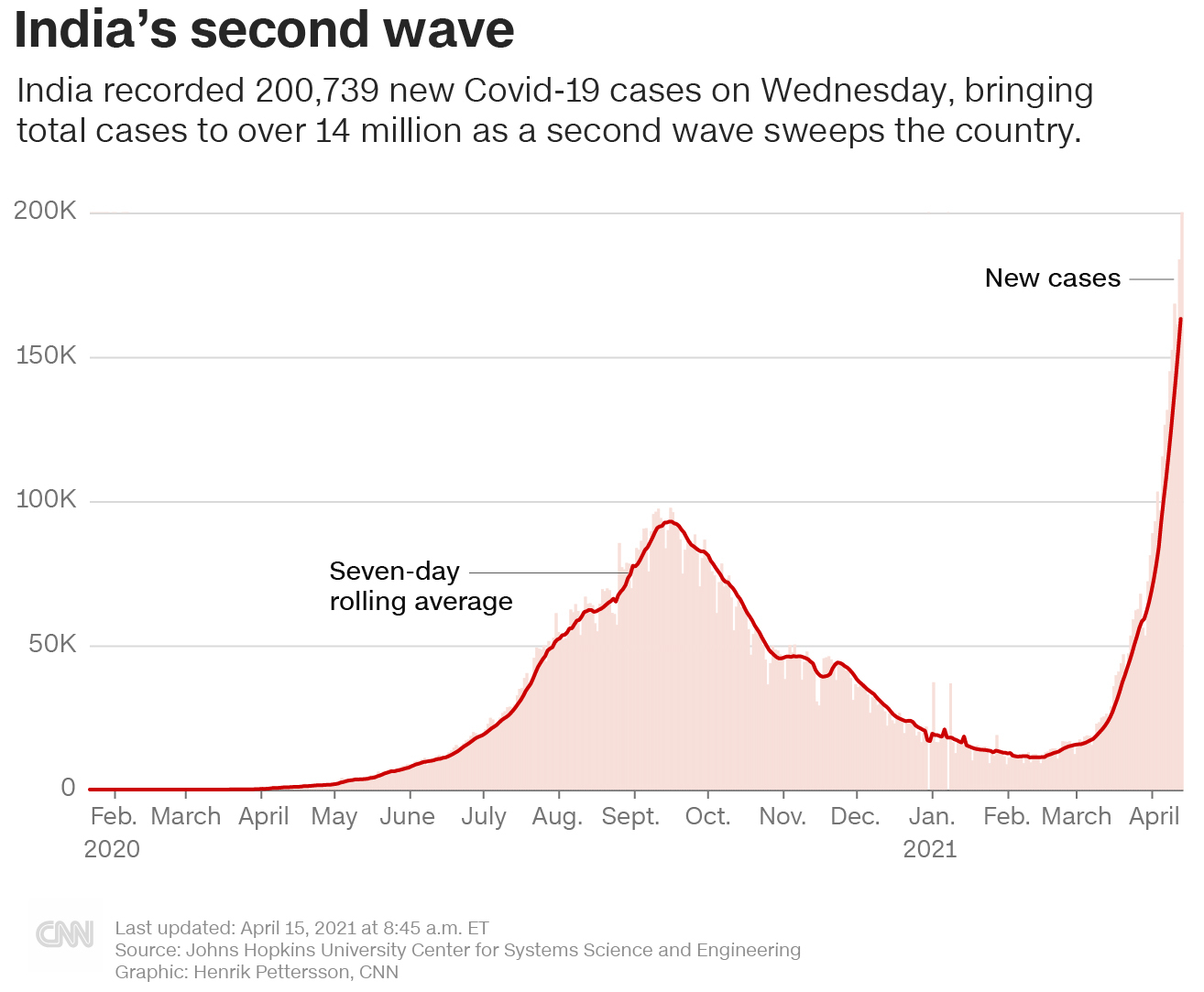
Experts call for travel restrictions as COVID-19 cases surge in India – CBS News
The coronavirus is spreading much faster in India than in any other country in the world, worrying experts about the threat posed to the rest of the world.
India has been reporting an average of nearly 150,000 new cases daily over the past week. On Tuesday, the country reported nearly 162,000 new infections — slightly lower than an all-time high of 168,912 fresh cases from a day earlier, raising the total number of cases in the country to nearly 13.6 million.
Experts believe the surge of infections in India poses a threat to other parts of the world because international travel hasn’t been significantly restricted.
“The high infection rate in India is worrying both because it indicates that in the absence of movement restrictions, and with significant vaccination, the virus continues to pose a serious threat,” Dr. Ramanan Laxminarayan, founder and director of the Washington-based Center for Disease Dynamics, Economics & Policy told CBS News.
“But because of India’s size, it is unlikely that the world would be done with Covid unless we are able to stop the epidemic here.”
India is now only behind the U.S., which has reported more than 31 million cases. Brazil at number three has 13.4 million cases.
Chloroquine is a potent inhibitor of SARS coronavirus infection and spread – National Institute of Health
[editor’s note: a reader pointed this out to me. I continue to believe that Chloroquine got politicized. The hyperlink above will send you to the NIH webpage ]
Vax Passports Are a Bad Idea – MedPage
There are many venues where a vaccine passport can be used. It can be used to restrict international travel. Already, some countries require proof of yellow fever vaccination to enter. It can be used to restrict access to unique shared spaces. When I attended college, proof of meningococcus vaccination was needed to live in the dorms. Some proponents of the SARS-CoV-2 vaccine passport envision it will be used beyond these precedents. They imagine the passport will be used to restrict access to daily activities with many participants such as movie theaters, malls, amusement parks, concerts, bars, and churches. In what follows, I will focus on this potential use of passports: regulating daily, domestic activities – this is fundamentally different than other forms of restriction, as it is a novel restriction.
… When it comes to daily domestic activities, a vaccine passport results in a trade-off. It very modestly changes the risk to vaccinated individuals and those who do not have a vaccine option. It changes the places unvaccinated individuals can congregate, which may or may not curb or fuel viral spread, depending on what they do instead. It will be met with political opposition and may result in unanticipated, unpredictable, harmful outcomes, including perpetuating discrimination. It will, at times, surely malfunction and some people will be angered by that. It will require massive political capital to institute.
In contrast with what I see – superficial analyses of the passport – my analysis is that they are, on balance, if used for the purposes I describe, a bad idea, and we should work to promote vaccination without this tool. Vaccination is good; vaccine passports for domestic activities are unrealistic and possibly unhelpful. That’s the sort of nuance we need more of in this pandemic.
Vinay Prasad, MD, MPH, is a hematologist-oncologist and associate professor of medicine at the University of California San Francisco, and author of Malignant: How Bad Policy and Bad Evidence Harm People With Cancer.
[editor’s note: there is a lot of meat in this post and deserves a full read]
Doubtful COVID-19 vaccine makers can provide enough shots for all U.S. adults, despite their commitments to ship more than a billion doses. – Government Accountability Office
The federal government has taken several actions to increase the availability of COVID-19 vaccine doses and indicated it expects to have enough doses available for all adults in the United States by the end of May. As of April 1, 2021, the government had purchased 1.2 billion doses of one- and two-dose regimen vaccines. Also, vaccine companies reported making additional manufacturing sites operational, among other actions to expand capacity and mitigate challenges.
Federal officials said projecting future availability of vaccine doses can be difficult, in part because of uncertainty surrounding complex manufacturing processes. Given this uncertainty, coupled with the significant manufacturing and distribution increases needed to have enough vaccine doses available for all adults, managing public expectations is critical. GAO’s prior work has found that timely, clear, and consistent communication about vaccine availability is essential to ensure public confidence and trust, especially as initial vaccine implementation did not match expectations.
Stakeholders GAO interviewed identified challenges with initial COVID-19 vaccine implementation. For example, some stakeholders said states often did not have information critical to distribution at the local level, such as how many doses they would receive and when. The federal government has begun initiatives—outlined in a national response strategy—to improve implementation, such as creating new vaccination sites. In its March 2021 distribution strategy, CDC provided a high-level description of its activities and noted that more details would be included in future reports to Congress. To meet the expectations set by recent announcements, such as the planned expansion of vaccine eligibility to all adults and the introduction of tools to help individuals find vaccines, it will be imperative that the federal government effectively coordinate and communicate its plans, as GAO recommended in September 2020.
[editor’s note: compare what you read here to the next post]
US could have 300 million excess vaccine doses by the end of July, report says – CNN
The US could have an estimated 300 million excess Covid-19 vaccine doses by the end of July, according to a report from Duke University.
Researchers used data on the US government’s advance purchase commitments with drug giants to arrive at the estimate.
The country has commitments for vaccines with Pfizer, Moderna, Johnson & Johnson, AstraZeneca and Novavax.
The AstraZeneca and Novavax shots are yet to receive emergency use authorization in the US, according to the Food & Drug Administration (FDA).
The report authors also reviewed vaccine production timelines and used US Census data to estimate demand.
Their estimate accounts for the nation retaining enough doses for most children in the country.
The researchers also assume that 75% of the US population will receive a two-dose vaccine and 25% will receive a single shot Johnson & Johnson vaccine.
Given the recent pause that will limit use of the Johnson & Johnson vaccine in the US, their projections may not be entirely accurate.
IBM flags more cyber attacks on COVID vaccine infrastructure – Reuters
International Business Machines Corp said on Wednesday its cybersecurity unit has uncovered more digital attacks targeting the global COVID-19 vaccine supply chain since the issue was originally flagged late last year.
The cloud services provider said it recently found that the phishing campaign has targeted 44 more companies which are involved in the complex logistical work of distribution of COVID-19 vaccines in 14 countries.
The campaign is targeting organizations associated with the COVID-19 vaccine “cold chain” – the process needed to keep vaccine doses at extremely cold temperatures as they travel from manufacturers to recipients.
IBM said some of the mails from hackers were sent several months in advance of the approval of any vaccine variant. It has previously said hackers were using meticulously crafted booby-trapped emails sent in the name of an executive with Haier Biomedical, a Chinese cold chain provider.
New Side Effect From mRNA COVID Vaccines? – MedPage
Herpes zoster reactivation — a.k.a. shingles — following COVID-19 vaccination in six patients with comorbid autoimmune/inflammatory diseases may be a new adverse event associated with the Pfizer/BioNTech mRNA vaccine, suggested a new report.
At two centers in Israel, there have been six cases of herpes zoster developing shortly after administration of the Pfizer vaccine in patients with disorders such as rheumatoid arthritis since December 2020, according to Victoria Furer, MD, of Tel Aviv University, and colleagues.
Little has been known about the safety and efficacy of the COVID-19 vaccines among patients with rheumatic diseases, because immunosuppressed individuals were not included in the initial clinical trials, they explained.
Accordingly, an observational study has been conducted at the Tel Aviv Medical Center and the Carmel Medical Center in Haifa, monitoring post-vaccination adverse events in patients with rheumatoid arthritis, spondyloarthropathies, connective tissue diseases, vasculitis, and myositis. This interim analysis included 491 patients and 99 controls, with events reported during the 6-week post-vaccination monitoring period. The prevalence among patients was 1.2% versus none in controls, Furer and colleagues reported in Rheumatology.
“We haven’t seen any additional cases so far,” Furer told MedPage Today. However, “further surveillance of potential adverse effects following anti-COVID-19 vaccination in patients with rheumatic diseases is warranted,” she added.
Merck to continue tests of Covid pill, but stop trial in hospitalized patients – STAT
Merck said Thursday that it has seen “encouraging” results in a clinical trial of an antiviral pill to treat Covid-19 early in the disease’s course, but the pill failed to help hospitalized patients and must be used very early in the disease.
At the same time, the company said it will stop efforts to develop a second medicine for patients who have already been hospitalized with the disease.
The drug giant licensed the anti-Covid pill, molnupiravir, from Ridgeback Biotherapeutics last July. In November, Merck spent $425 million to buy OncoImmune, in order to obtain an immune-modifying treatment that already seemed to have a benefit for patients hospitalized with Covid-19.
Roy Baynes, the chief medical officer at Merck Research Laboratories, told STAT that proving the OncoImmune drug’s benefit to regulators would require another study, and that there is still work to be done on manufacturing the therapy. “Given where we are, we feel we can bring more value focusing on the full development of molnupiravir,” Baynes said.
Baynes said that a 302-patient study of molnupiravir in people with Covid-19 who have not been hospitalized has shown signs of benefit for the drug. The percentage of patients who were hospitalized or died in the treatment groups was lower than in the placebo group, but Merck said “the number of events reported are not sufficient to provide a meaningful measure of clinical effect.” Baynes said the medicine showed “a very clear antiviral effect.” Actual data will be presented at a medical meeting.
Phase 3 trial finds corticosteroid inhaler speeds recovery time in COVID-19 patients – News-Medical
The corticosteroid budesonide shows promise in treating patients with mild COVID-19 infection. Interim analysis of the phase 3 PRINCIPLE clinical trial was posted to the medRxiv* preprint server. Researchers found a 3-day faster recovery of COVID-19 symptoms in people at high risk for severe side effects administered a budesonide inhaler. There was also a 2.1% reduction in hospitalization and death.
Corticosteroids such as dexamethasone have shown to be effective in alleviating the severity of COVID-19 infection. Adding budesonide could give doctors more options in treating patients, especially as it is already readily available in many primary care settings and is in the World Health Organization’s List of Essential Medicines. The medicine is also known to be easier to use for unwell, comorbid, and potentially frail older patients.
The research team writes:
“Several randomized trials have demonstrated the benefits of systemic corticosteroids for the treatment of people hospitalized with COVID-19. Our findings are immediately relevant for clinical practice as they suggest that early treatment in the community with inhaled corticosteroids is effective at speeding recovery, which has important benefits for patients and wider society.”
The trial is still ongoing, with the final analysis being made.
Vaccines Carry Far Lower Risk for Rare Blood Clots Than COVID, Study Shows – Medscape
The risk of developing cerebral venous thrombosis (CVT) from COVID-19 was “many-fold” higher than from receiving the AstraZeneca/Oxford or the mRNA vaccines from Pfizer and Moderna, researchers have concluded.
A preprint study by the University of Oxford found that from a dataset of over 500,000 COVID patients, CVT would have occurred in 39 per million people.
CVT has been reported to occur in about 5 per million people after a first dose of the AstraZeneca/Oxford vaccine. In over 480,000 people receiving either the Pfizer/BioNTech or Moderna mRNA vaccines, CVT occurred in 4 per million.
The researchers said that compared to the mRNA vaccines, the risk of CVT from COVID-19 was about 10 times greater.
Compared to the Oxford vaccine, the risk of CVT from COVID-19 was about 8 times greater.
The COVID vaccine from Johnson & Johnson, which has been put into limbo because of cases of rare blood clots in patients that received it, is an adenovirus vaccine similar to AstraZeneca’s but was not included in this research.
A similar pattern was seen in portal vein thrombosis (PVT) which occurred in 436.4 per million people who had COVID. That compared to 44.9 per million for the mRNA vaccine group, and 1.6 per million for those receiving the Oxford vaccine.
Pfizer CEO: Third dose of COVID-19 vaccine ‘likely’ needed within 1 year – The Hill
Pfizer’s CEO said Thursday that COVID-19 vaccine recipients will “likely” need a third dose between six to 12 months after they’re fully vaccinated and suggested vaccinations for coronavirus could be needed every year.
Albert Bourla told CNBC’s Bertha Coombs at a CVS Health event that he predicts based on current data a “likely scenario” will involve the COVID-19 vaccine being administered to patients annually.
Bourla said it “remains to be seen” how often any potential additional vaccines would be provided.
“A likely scenario is there will be likely a need for a third dose somewhere between six and 12 months and then from there there would be an annual revaccination,” he said at the event.
“But all of that needs to be confirmed and again the variants will play a key role,” he continued. “It is extremely important to suppress the pool of people that can be susceptible to the virus because they are vaccinated with high-efficacy vaccines.”
Earlier this month, Pfizer, along with German partner BioNTech, reported their vaccine remained 91 percent effective at least six months after the second dose.
US is making plans just in case the Covid-19 vaccines need a booster, officials say – CNN
The US is making plans just in case Covid-19 vaccines need booster doses later, officials told a congressional hearing Thursday.
“We are planning for potential booster doses of vaccines, if they are needed,” Dr. David Kessler, chief science officer for the Biden administration’s Covid response, told a hearing of the House Select Subcommittee on the Coronavirus Crisis. “As with other vaccines, a subsequent dose may be desirable.”
Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said that there were a few different approaches that could boost the potency of a Covid-19 vaccine.
One is to create a booster that would strengthen the original vaccine and would be strong enough to protect against variants. The other is to make a booster that would work against particular variants. While the variant first found in South Africa, known as B.1.351, is not dominant in the US, it theoretically could be a bigger problem for the existing vaccines, and vaccine makers could target that particular variant.
“The problem with that, is, that if you get more and more variants, that’s almost like playing Whack-a -mole,” Fauci said.
“You hit this one, then you go to the other one, you go to the other one, and that’s the reason why, what we’re putting a lot of effort in, is to try and get a more universal vaccine that would cover all different types of variants. That’s the ultimate end game,” Fauci said.
He added that research is already underway. Until then, scientists are trying to figure out what the most dangerous variant is and to make a special boost against that.
The following are foreign headlines with hyperlinks to the posts
Olympics could be canceled because of virus, Japan ruling party figure admits
A top politician in Japan talked of canceling the Olympics. Then he walked it back.
Western concerns over the Johnson & Johnson and AstraZeneca vaccines are jeopardizing inoculation campaigns around the world.
“Collapse is imminent” for São Paolo hospitals buckling under Brazil’s ongoing COVID crisis, raising the heat on President Jair Bolsonaro.
EU Commission to End AstraZeneca, J&J Vaccine Contracts at Expiry
CanSinoBIO Says No Serious Blood Clots From Its Vaccine
Trudeau comes under fire as vaccine shortages hit Canadian clinics.
Experts Think France’s COVID Death Total Underestimated by Thousands
New Delhi Converting a Dozen Large Buildings Into COVID Treatment Centers
Indian capital imposes weekend curfew as cases rise
As India logs 200,000 daily infections, a new exodus from cities has begun.
France Covid-19 death toll surpasses 100,000
Norway will stop using the AstraZeneca Covid-19 vaccine as part of its vaccination program
WHO: Europe surpasses one million Covid-19 deaths
The following additional national and state headlines with hyperlinks to the posts
Moderna said it will need more than a few months to further expand manufacturing capacity for its vaccine.
Deaths from drug overdoses surged in the U.S. last year, as the pandemic drained treatment resources.
Cosmopolitan hotel-casino promises $1M in cash bonuses if employees vaccinated by May 1
What Is behind the Global Jump in Personal Saving during the Pandemic?
India Breaches 200,000 Daily COVID-19 Cases as Hospital Beds, Oxygen Fall Short
In a phase III trial, nitazoxanide did not improve recovery time in mild or moderate COVID-19, but it did slash the progression to severe illness, drugmaker Romark announced.
Big surprise: airlines push back against a new CDC study showing substantially less risk of SARS-CoV-2 transmission when the middle seat is left empty.
Penn study suggests those who had COVID-19 may only need one vaccine dose
With impressive accuracy, dogs can sniff out coronavirus
More than half of COVID-19 patients report persistent symptoms 12-months after infection
One in 4 Americans would refuse a coronavirus vaccine if offered, a recent NPR/Marist poll found. Another 5% are “undecided” about whether they would get the shot. And some researchers are growing worried that this reluctance will be enough to prevent the nation from reaching what’s known as herd immunity.
CDC finds less than 1 percent of fully vaccinated people got COVID-19
Cancer Patient’s Tumors Shrink After Having Severe COVID, Doctors Find
New coronavirus tests should still be able to detect variants, but health officials are vigilant.
Today’s Posts On Econintersect Showing Impact Of The Pandemic With Hyperlinks
10 April 2021 New York Fed Weekly Economic Index (WEI): Index Again Improved
February 2021 Business Inventories Remain Normal For Times Of Economic Expansion
March 2021 Headline Industrial Production Improves
April 2021 Philly Fed Manufacturing Survey Index Improvement Continues
April 2021 Empire State Manufacturing Index Strongly Improves
Headline Retail Sales Skyrocket in March 2021
10 April 2021 Initial Unemployment Claims Rolling Average Improves
Documentary Of The Week: Historical Perspectives For The COVID-19 Pandemic
Johnson And Johnson Vaccine Suspension: A Doctor Explains What This Means For You
Warning to Readers
The amount of politically biased articles on the internet continues to increase. And studies and opinions of the experts continue to contradict other studies and expert opinions. Honestly, it is difficult to believe anything anymore.

I assemble this coronavirus update daily – sifting through the posts on the internet. I try to avoid politically slanted posts (mostly from CNN, New York Times, and the Washington Post) and can usually find unslanted posts on that subject from other sources on the internet. I wait to publish posts on subjects that I cannot validate across several sources. But after all this extra work, I do not know if I have conveyed the REAL facts. It is my job to provide information so that you have the facts necessary – and then it is up to readers to draw conclusions.
Analyst Opinion of Coronavirus Data
There are several takeaways that need to be understood when viewing coronavirus statistical data:
- The global counts are suspect for a variety of reasons including political. Even the U.S. count has issues as it is possible that as much as half the population has had coronavirus and was asymptomatic. It would be a far better metric using a random sampling of the population weekly. In short, we do not understand the size of the error in the tracking numbers.
- Just because some of the methodology used in aggregating the data in the U.S. is flawed – as long as the flaw is uniformly applied – you establish a baseline. This is why it is dangerous to compare two countries as they likely use different methodologies to determine who has (and who died) from coronavirus.
- COVID-19 and the flu are different but can have similar symptoms. For sure, COVID-19 so far is much more deadly than the flu. [click here to compare symptoms]
- From an industrial engineering point of view, one can argue that it is best to flatten the curve only to the point that the health care system is barely able to cope. This solution only works if-and-only-if one can catch this coronavirus once and develops immunity. In the case of COVID-19, herd immunity may need to be in the 80% to 85% range. WHO warns that few have developed antibodies to COVID-19 when recovering from COVID-19. Herd immunity does not look like an option without immunization although there is now a discussion of whether T-Cells play a part in immunity [which means one might have immunity without antibodies]
- Older population countries will have a significantly higher death rate as there is relatively few hospitalizations and deaths in younger age groups..
- There are at least 8 strains of the coronavirus. California and New York may have a deadlier strain imported from Europe, compared to less deadly viruses elsewhere in the United States.
- Each publication uses different cutoff times for its coronavirus statistics. Our data uses 11:00 am London time. Also, there is an unexplained variation in the total numbers both globally and in the U.S.
What we do or do not know about the coronavirus [actually there is little scientifically proven information]. Most of our knowledge is anecdotal, from studies with limited subjects, or from studies without peer review.
- How many people have been infected as many do not show symptoms?
- Masks do work. Unfortunately, early in the pandemic, many health experts — in the U.S. and around the world — decided that the public could not be trusted to hear the truth about masks. Instead, the experts spread a misleading message, discouraging the use of masks.
- Current thinking is that we develop at least 5 months of immunity from further COVID infection.
- The Moderna and Pfizer vaccines have an effectiveness rate of about 95 percent after two doses. That is on par with the vaccines for chickenpox and measles. The 95 percent number understates the effectivenessas it counts anyone who came down with a mild case of Covid-19 as a failure. But turning Covid into a typical flu — as the vaccines evidently did for most of the remaining 5 percent — is actually a success. Of the 32,000 people who received the Moderna or Pfizer vaccine in a research trial, only one contracted a severe Covid case.
- To what degree do people who never develop symptoms contribute to transmission? Research early in the pandemic suggested that the rate of asymptomatic infections could be as high as 81%. But a meta-analysis, which included 13 studies involving 21,708 people, calculated the rate of asymptomatic presentation to be 17%.
- The accuracy of rapid testing is questioned – and the more accurate test results are not being given in a timely manner.
- Can children widely spread coronavirus? [current thinking is that they are a minor source of the pandemic spread]
- Why have some places avoided big coronavirus outbreaks – and others hit hard?
- Air conditioning contributes to the pandemic spread.
- It appears that there is increased risk of infection and mortality for those living in larger occupancy households.
- Male patients have almost three times the odds of requiring intensive treatment unit (ITU) admission compared to females.
- Outdoor activities seem to be a lower risk than indoor activities.
Treatments with solid scientific support:
- Dexamethasone
- Proning, or turning someone on their stomach
- Remdesivir
- Baricitinib
Treatments with potential but limited evidence:
- ECMO, or extracorporeal membrane oxygenation
- fluvoxamine
- Cyclosporine
- Famotidine
- Intravenous immunoglobulin
- Ivermectin
- Interferons
Drugs shown to be ineffective:
- The combination of lopinavir-ritonavir
- Hydroxychloroquine
- Insulin
- High dose zinc and vitamin C
- Convalescent plasma
- Monoclonal antibodies
- Tocilizumab
- Anti-coagulants
- A current scientific understanding of the way the coronavirus works can be found [here].
There is now a vaccine available – the questions remain:
- will there be any permanent side effects that will appear months from now,
- how long immunity will last [we can currently say we do not know if it will last more than 4 months],
- there is no solid evidence yet the vaccine will block transmission
Heavy breakouts of coronavirus have hit farmworkers. Farmworkers are essential to the food supply. They cannot shelter at home. Consider:
- they have high rates of respiratory disease [occupational hazard]
- they travel on crowded buses chartered by their employers
- few have health insurance
- they cannot social distance and live two to four to a room – and they eat together
- some reports say half are undocumented
- they are low paid and cannot afford not to work – so they will go to work sick
- they do not have access to sanitation when working
- a coronavirus outbreak among farmworkers can potentially shutter entire farm
The bottom line is that COVID-19 so far has been shown to be much more deadly than the data on the flu. Using CDC data, the flu has a mortality rate between 0.06 % and 0.11 % Vs. the coronavirus which to date has a mortality rate of 4 % [the 4% is the average of overall statistics – however in the last few months it has been hovering around 1.0%] – which makes it between 10 and 80 times more deadly. The reason for ranges:
Because influenza surveillance does not capture all cases of flu that occur in the U.S., CDC provides these estimated ranges to better reflect the larger burden of influenza.
There will be a commission set up after this pandemic ends to find fault [it is easy to find fault when a once-in-a-lifetime event occurs] and to produce recommendations for the next time a pandemic happens. Those that hate President Trump will conclude the virus is his fault.
Resources:
- Get the latest public health information from CDC: https://www.coronavirus.gov .
- Get the latest research from NIH: https://www.nih.gov/coronavirus.
- Find NCBI SARS-CoV-2 literature, sequence, and clinical content: https://www.ncbi.nlm.nih.gov/sars-cov-2/.
- List of studies: https://icite.od.nih.gov/covid19/search/#search:searchId=5ee124ed70bb967c49672dad
include(“/home/aleta/public_html/files/ad_openx.htm”); ?>







