Written by Steven Hansen
The U.S. new cases 7-day rolling average are 18.0 % LOWER than the 7-day rolling average one week ago and U.S. deaths due to coronavirus are now 6.5 % LOWER than the rolling average one week ago. Today’s posts include:
- U.S. Coronavirus New Cases are 47,430
- U.S. Coronavirus deaths are at 479
- U.S. Coronavirus immunizations have been administered to 69 doses per 100 people.
- The 7-day rolling average rate of growth of the pandemic shows new cases worsened and deaths improved
- It was rare but possible for COVID-19 patients without cardiovascular disease (CVD) or cardiovascular risk factors to develop new heart failure (HF) during their illness
- Triple mutation in SARS-CoV-2 seen in second wave of COVID-19 in India
- Hepatitis C drugs boost Remdesivir’s antiviral activity against COVID-19
- Research suggests that Ginkgo biloba extract contains naturally occurring inhibitors against SARS-CoV-2
- A new treatment that might keep COVID-19 patients off the ventilator
- Lots of additional headlines.

include($_SERVER[‘DOCUMENT_ROOT’].’/pages/coronavirus1.htm’); ?>
Hospitalizations Are The Only Accurate Gauge
Hospitalizations historically appear to be little affected by weekends or holidays. The hospitalization growth rate trend is improving.
source: https://gis.cdc.gov/grasp/covidnet/COVID19_3.html
Historically, hospitalization growth follows new case growth by one to two weeks.
As an analyst, I use the rate of growth to determine the trend. But, the size of the pandemic is growing in terms of real numbers – and if the rate of growth does not become negative – the pandemic will overwhelm all resources.
The graph below shows the rate of growth relative to the growth a week earlier updated through today [note that negative numbers mean the rolling averages are LOWER than the rolling averages one week ago]. As one can see, the rate of growth for new cases peaked in early December 2020 for Thanksgiving, and early January 2021 for end-of-year holidays – and it now shows that the coronavirus effect is lower.
In the scheme of things, new cases decline first, followed by hospitalizations, and then deaths.
The New Variants Are The Primary Cause Of This Fourth Wave
Even with vaccinations picking up, the fourth wave is now underway.
- the more people that are vaccinated reduces the pool of people that can be infected. Today we have removed over 69 % of the population from being infected which theoretically should reduce the infection rate by 69 % [it is unproven whether the vaccines prevent a vaccinated person from being a carrier of the virus even though showing no signs]. If the vaccines are shown to stop transmission, then in theory it would reduce the infection rate by double the percent vaccinated [in this case you prevent your own infection and do not pass it along to another].
- it is also unknown what the effective rate of the current vaccines is against mutations that seem to appear almost daily. As an example, if the effective rate drops to 60%, it means the 69 % reduction in the infection rate discussed above is almost cut in half. The South African and Brazilian variant is reported somewhat immune to the current vaccines.
- In theory, the pandemic should be over immediately if everyone could be vaccinated today. The problem is that every day brings a new mutation (which would not appear if the pandemic was stopped). The longer the immunization process takes – the more ineffective the vaccine will become.
- It is not clear whether the vaccine prevents those vaccinated from spreading the virus. It seems to be well documented that it normally stops the virus from taking hold and when it does not – the infection is mild.
The real question is to what extent the vaccines will be mitigating this surge.
Coronavirus News You May Have Missed
CDC Throws Open the Doors on Outdoor Masking – MedPage
People vaccinated against COVID-19 are no longer required to wear masks outdoors in most settings, according to updated guidance released by the CDC on Tuesday.
Fully vaccinated people can now “gather or conduct activities outdoors without wearing a mask, except in certain crowded settings or venues.” Safe activities include being outdoors with members of your household, attending small outdoor gatherings with vaccinated and unvaccinated people, and dining outdoors at a restaurant with friends from multiple households.
While the agency graded attending a crowded outdoor event like a live performance, parade, or sporting event as “safe” for vaccinated individuals, they still recommend masking during these events.
CDC updated masking guidance for unvaccinated individuals as well — noting that even those who are unvaccinated can now “walk, run or bike outdoors with members of your household” and an unvaccinated individual can “attend a small, outdoor gathering with fully vaccinated family and friends” without a face covering.
But unvaccinated people should continue to wear masks when attending small outdoor gatherings with a mix of fully vaccinated and unvaccinated people, though CDC grades this activity as “safe.” They should also continue to wear masks when dining outdoors at a restaurant with friends from multiple households (graded as “less safe”) and when attending crowded outdoor events (graded as “least safe”).
At a briefing on Tuesday, CDC director Rochelle Walensky, MD, emphasized that while outdoor activities without a mask are safe for vaccinated people, “we continue to recommend masking in crowded outdoor settings and venues, such as packed stadiums and concerts, where there is decreased ability to maintain physical distance and where many unvaccinated people may also be present. We will continue to recommend this until widespread vaccination is achieved.”
[editor’s note: also read When do we have to wear a mask outside? The C.D.C. says less often, and here’s how and why.]
Can Heart Failure Show Up in COVID Patients Out of the Blue? – MedPage
It was rare but possible for COVID-19 patients without cardiovascular disease (CVD) or cardiovascular risk factors to develop new heart failure (HF) during their illness, according to a center in New York.
A retrospective analysis of 6,439 hospitalized COVID-19 patients found that 0.6% had new HF and 6.6% had a history of HF, reported Anuradha Lala, MD, of Icahn School of Medicine at Mount Sinai in New York City, and colleagues.
As shown in their research letter online in the Journal of the American College of Cardiology, out of the 37 people who experienced new HF, 13 had presented with shock (four cardiogenic, six septic, three mixed) and five with acute coronary syndrome. Just eight had neither CVD nor any related risk factors, whereas 14 had a history of CVD and another 15 at least one risk factor.
The eight individuals developing new HF despite a lack of CVD tended to be younger and have a lower body mass index and fewer comorbidities compared with other new HF patients.
These eight had similar lengths of stay in the hospital as peers with existing CVD or cardiovascular risk factors. However, these patients were more likely to require intensive care admission and intubation, and had a lower risk of in-hospital mortality, Lala’s group reported.
“As viral illnesses such as influenza have been reported to precipitate new HF, similar speculative correlations have been drawn with COVID-19. To the best of our knowledge, this is the largest study to date to provide a context for reports of new-onset HF in the setting of hospitalization for SARS-CoV2 infection,” the authors said.
“We need to learn more about how SARS-CoV-2 … may directly affect the cardiovascular system and precipitate new HF — as to whether it is an indirect effect of critical illness or direct viral invasion,” Lala said in a Mount Sinai press release.
“Importantly, though symptoms of HF — namely shortness of breath — can mimic symptoms associated with COVID-19, being alerted to the findings of this study may prompt clinicians to monitor for signs of congestion more consistent with HF than COVID-19 alone,” she continued.
A new treatment that might keep COVID-19 patients off the ventilator – EurekAlert
A new treatment is among the first known to reduce the severity of acute respiratory distress syndrome caused by the flu in animals, according to a new study.
Tests in mice infected with high doses of influenza showed that the treatment could improve lung function in very sick mice and prevent progression of disease in mice that were pre-emptively treated after being exposed to the flu.
The hope is that it may also help humans infected with the flu, and potentially other causes of acute respiratory distress syndrome (ARDS) such as SARS-CoV-2 infection.
Specific cells in mice are less able to make key molecules after influenza invades the lungs, reducing their ability to produce a substance called surfactant that enables lungs to expand and contract. The shortage of surfactant is linked to ARDS, an illness so serious that it typically requires mechanical ventilation in an ICU.
Researchers bypassed the blocked process in mice by re-introducing the missing molecules alone or in combination as an injected or oral treatment. The results: normalized blood oxygen levels and reduced inflammation in mouse lungs – effects that could make a person well enough for hospital discharge.
“The most important and impressive thing in this study is the fact that we have benefits even when we treat late in the disease process. If we could develop a drug based on these findings, you could take somebody who’s going to have to go on a ventilator and stop that completely,” said Ian Davis, professor of veterinary biosciences at The Ohio State University and senior author of the study. “There’s nothing out there now that can do this for ARDS that will bring them back to that degree, and certainly not for flu.”
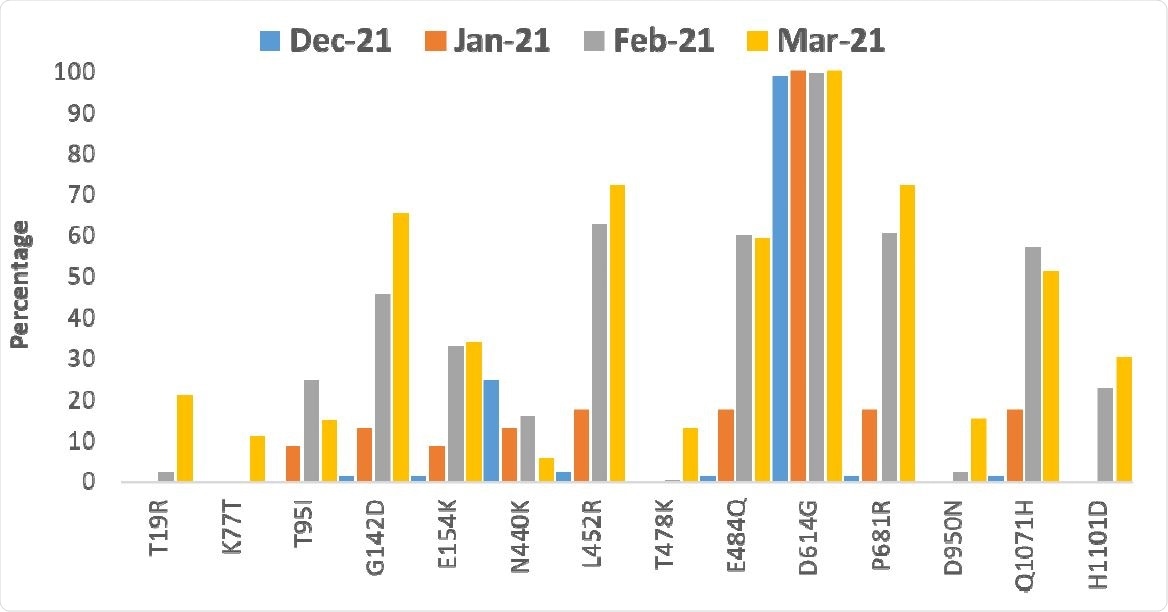
Triple mutation in SARS-CoV-2 seen in second wave of COVID-19 in India – News-Medical
Researchers sequenced the viral genome from samples in the state of Maharashtra and found a unique combination of three mutations that suggest the SARS-CoV-2 virus is continually evolving to evade the human immune response.
With the COVID-19 pandemic continuing to spread in several parts of the world, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is evolving to evade our immune system. Several new variants, designated variants of concern, have been reported from the United Kingdom, South Africa, and Brazil at the end of 2020, which seem to be more infectious than the original strain.
The introduction of new variants and their consequences is important in guiding public health response in a country. With the tremendous spike in COVID-19 seen in India, especially in the state of Maharashtra, the government increased the sequencing of the viral genome to identify possible new mutants and understand the fitness of new strains.
Researchers from several Indian institutes involved in SARS-CoV-2 research obtained samples from international travelers to Maharashtra. About 5% of the surveillance samples tested positive and were sequenced, and the whole genomes of SARS-CoV-2 analyzed.
The team also analyzed the crystal structure of the top 10 virus spike protein mutations complexed with the human receptor, angiotensin-converting enzyme 2 (ACE2). They also assessed the effect of mutations on binding to two neutralizing antibodies using the structures.
Trend of major mutations in the spike protein from December, 2020 to March, 2021.
Pfizer CEO: Oral drug to stop coronavirus could be ready next year – The Hill
Pfizer CEO Albert Bourla says that an oral drug for COVID-19 could be ready next year.
“If all goes well, and we implement the same speed that we are, and if regulators do the same, and they are, I hope that by the end of the year,” Bourla said on CNBC’s “Squawk Box.”
The only antiviral currently approved for use against the coronavirus is remdesivir, which is manufactured by Gilead Sciences. The Food and Drug Administration (FDA) gave the drug full approval in October, after it was granted emergency use authorization in May.
Remdesivir is administered in a hospital setting through an IV.
Speaking on “Squawk Box,” Bourla said Pfizer is working on two antivirals, an oral and an injectable.
The company began an early-stage clinical trial of the oral drug late last month. The drug blocks protease, a critical enzyme that the virus needs to replicate.
“Particular attention is on the oral because it provides several advantages,” Bourla said. “One of them is that you don’t need to go to the hospital to get the treatment, which is the case with all the injectables so far. You could get it at home, and that could be a game-changer.”
He also said the medication could be “way more effective against the multiple variants” of the virus than current options.
Bourla said the company should have more news about it later this summer.
Hepatitis C drugs boost Remdesivir’s antiviral activity against COVID-19 – EurekAlert
Remdesivir is currently the only antiviral drug approved in the U.S. for treating COVID-19 patients. In a paper published this week in Cell Reports, researchers from The University of Texas at Austin, Rensselaer Polytechnic Institute (RPI) and the Icahn School of Medicine at Mount Sinai showed that four drugs used to treat hepatitis C render remdesivir 10 times better at inhibiting the coronavirus in cell cultures.
These results indicate that a mixture containing remdesivir and a repurposed hepatitis C virus (HCV) drug could potentially function as a combination antiviral therapy for SARS-CoV-2. Such an antiviral could provide an immediate treatment for unvaccinated people who become infected and for vaccinated people whose immunity has waned.
Because these hepatitis drugs are already approved for use and their potential side effects are known, such a combination therapy could be tested in humans more quickly than for a new drug. One big drawback with remdesivir, however, is that it must be administered intravenously, limiting its use to patients already admitted to the hospital.
“Our goal is to develop a combination of oral drugs that can be administered to outpatients before they are sick enough to require hospitalization,” said Robert M. Krug, professor emeritus of molecular biosciences at UT Austin and co-author of the paper. “The HCV drugs that enhance remdesivir’s antiviral activity are oral drugs. Ideally, we would need an oral drug that inhibits SARS-CoV-2 in the same way as remdesivir to develop an effective combination treatment.”
Research suggests that Ginkgo biloba extract contains naturally occurring inhibitors against SARS-CoV-2 – News-Medical
The coronavirus disease (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), further spreads across the globe, causing skyrocketing cases in some countries.
Finding effective agents against the virus is crucial to combat the current global health crisis. Past evidence has shown that Gingko Biloba, also known as maidenhair, can inhibit viral proteins, inhibiting replication.
Researchers at Shihezi University in China found that Gingko Biloba leaves extract contains the most potent SARS-CoV-2 3-Chymotrypsin-like protease (3CLpro). This virally encoded main proteinase is essential for viral replication in coronaviruses.
Finding key active ingredients
It is well-known that the phytochemicals from medicinal plants are potential candidates. The study, published in the journal Fitoterapia, aims to discover the naturally occurring SARS-CoV-2 3CLpro inhibitors from herbal medicines. It also investigated the inhibitory mechanism of the newly found effective SARS-CoV-2 3CLpro inhibitors.
The researchers conducted a large-scale screening to determine biochemicals that may have antiviral activity, collecting 80 herbal products. They also used a biochemical approach to determine if these herbal products can inhibit SARS-CoV-2 3CLpro.
After the screening, they identified Gingko Biloba leaves extract (GBLE) as the most potent action against SARS-CoV-2 3CLpro. Further, the team wanted to determine the critical ingredients of GBLE responsible for inhibiting SARS-CoV-2.
A total of 20 natural ingredients were isolated from Gingko Biloba leaves. The researchers assayed these constituents to determine which ones are potential inhibitors, which can be used to mitigate the spread of COVID-19.
Of all the natural ingredients, bioflavonoids and ginkgolic acids (GAs) were observed to have the most potent inhibitory mechanisms on the 3CLpro molecules. Finding these active ingredients can help pave the way for developing novel anti-COVID-19 agents.
The following are foreign headlines with hyperlinks to the posts
The European Medicines Agency and European Centre for Disease Prevention and Control will launch observational studies to enhance post-marketing monitoring of the safety, effectiveness, and impact of COVID-19 vaccines.
In Maharashtra’s Nandurbar, a district with a population of over 16 lakh, the story of the second wave of coronavirus reads differently than the rest of the country. While India’s healthcare infrastructure finds itself overwhelmed by the surge in cases, it’s a pleasant surprise to see that in times like these, the district has 150 vacant beds and two oxygen plants that have a combined capacity to produce 2,400 litres per minute.
Turkey, which ranks fourth in the world in new daily cases per person, ordered a three-week lockdown.
In France, lockdown rules have become dizzyingly complex.
An imam established a COVID vaccination center at a British mosque and has been speaking out against vaccine disinformation, helping persuade his congregants.
With falling COVID-19 cases, Finland ended its state of emergency.
Indian Health Minister Says Nation Better Prepared For COVID Than in 2020
India Using Train Cars to Treat COVID Patients as Hospitals Run Out of Beds
India’s Election Commission bans state election victory processions as Covid-19 cases surge
Provinces in Pakistan ban non-urgent surgeries as oxygen supplies run low
Covid-19 cases are also surging in India’s neighboring Nepal
EU member states and Bhutan pledge to provide oxygen to India amidst crisis
Aid is coming to India, but its Covid-19 catastrophe is likely to continue.
Video of Oxygen Truck With Police Escort Shows India’s Dire COVID Situation
Biden says he intends to share coronavirus vaccine doses with India
Brazil rejects Russia’s Sputnik V coronavirus vaccine, citing safety concerns
Russia Says U.S. Pressured Brazil Not to Approve Sputnik COVID Vaccine
Israel study suggests elementary school reopenings are low-risk for COVID-19 transmission
France’s Sanofi to Help Moderna Manufacture COVID-19 Shots
COVID Treatment Has Improved, but Many Wish for an Easy Pill
Eyeing summer tourist season, Greece exempts more travelers from quarantine.
Irish health officials recommend the use of AstraZeneca and J&J vaccine for people over 50
Wimbledon hopes to have a “minimum” of 25% spectator capacity this year’s tournament
The following additional national and state headlines with hyperlinks to the posts
California ranked 50th among the states where coronavirus was spreading the fastest on a per-person basis, a USA TODAY Network analysis of Johns Hopkins University data shows.
Can you have alcohol after the Covid vaccine? (In short: probably, but not too much.)
Lawmakers in more than 40 states have introduced legislation that would forbid mandates requiring people get vaccinated.
Some public health experts say fully vaccinated people should continue masking when gathering in public.
Alaskan state Sen. Lora Reinbold (R) has been banned from flying Alaska Airlines for her “continued refusal to comply” with the airline’s mask mandate.
Exposure to high heat neutralizes SARS-CoV-2 in less than one second [72 deg C]
First cases of B.1.1.7 variant with E484K mutation in Pennsylvania
West Virginia Will Pay Young People $100 To Get Vaccinated Against COVID-19
Biden administration easing travel restrictions on international students
Burning Man festival canceled for a second straight year
US restaurant Covid-19 relief fund will open May 3
Today’s Posts On Econintersect Showing Impact Of The Pandemic With Hyperlinks
April 2021 Conference Board Consumer Confidence Nears Pre-Pandemic Levels.
April 2021 Richmond Fed Manufacturing Survey Unchanged
COVID-19 In Brazil: How Jair Bolsonaro Created A Calamity
New Malaria Vaccine Proves Highly Effective And COVID Shows How Quickly It Could Be Deployed
India Emerges As The World’s COVID-19 Hotspot
Warning to Readers
The amount of politically biased articles on the internet continues to increase. And studies and opinions of the experts continue to contradict other studies and expert opinions. Honestly, it is difficult to believe anything anymore.

I assemble this coronavirus update daily – sifting through the posts on the internet. I try to avoid politically slanted posts (mostly from CNN, New York Times, and the Washington Post) and can usually find unslanted posts on that subject from other sources on the internet. I wait to publish posts on subjects that I cannot validate across several sources. But after all this extra work, I do not know if I have conveyed the REAL facts. It is my job to provide information so that you have the facts necessary – and then it is up to readers to draw conclusions.
Analyst Opinion of Coronavirus Data
There are several takeaways that need to be understood when viewing coronavirus statistical data:
- The global counts are suspect for a variety of reasons including political. Even the U.S. count has issues as it is possible that as much as half the population has had coronavirus and was asymptomatic. It would be a far better metric using a random sampling of the population weekly. In short, we do not understand the size of the error in the tracking numbers.
- Just because some of the methodology used in aggregating the data in the U.S. is flawed – as long as the flaw is uniformly applied – you establish a baseline. This is why it is dangerous to compare two countries as they likely use different methodologies to determine who has (and who died) from coronavirus.
- COVID-19 and the flu are different but can have similar symptoms. For sure, COVID-19 so far is much more deadly than the flu. [click here to compare symptoms]
- From an industrial engineering point of view, one can argue that it is best to flatten the curve only to the point that the health care system is barely able to cope. This solution only works if-and-only-if one can catch this coronavirus once and develops immunity. In the case of COVID-19, herd immunity may need to be in the 80% to 85% range. WHO warns that few have developed antibodies to COVID-19 when recovering from COVID-19. Herd immunity does not look like an option without immunization although there is now a discussion of whether T-Cells play a part in immunity [which means one might have immunity without antibodies]
- Older population countries will have a significantly higher death rate as there is relatively few hospitalizations and deaths in younger age groups..
- There are at least 8 strains of the coronavirus. California and New York may have a deadlier strain imported from Europe, compared to less deadly viruses elsewhere in the United States.
- Each publication uses different cutoff times for its coronavirus statistics. Our data uses 11:00 am London time. Also, there is an unexplained variation in the total numbers both globally and in the U.S.
What we do or do not know about the coronavirus [actually there is little scientifically proven information]. Most of our knowledge is anecdotal, from studies with limited subjects, or from studies without peer review.
- How many people have been infected as many do not show symptoms?
- Masks do work. Unfortunately, early in the pandemic, many health experts — in the U.S. and around the world — decided that the public could not be trusted to hear the truth about masks. Instead, the experts spread a misleading message, discouraging the use of masks.
- Current thinking is that we develop at least 5 months of immunity from further COVID infection.
- The Moderna and Pfizer vaccines have an effectiveness rate of about 95 percent after two doses. That is on par with the vaccines for chickenpox and measles. The 95 percent number understates the effectivenessas it counts anyone who came down with a mild case of Covid-19 as a failure. But turning Covid into a typical flu — as the vaccines evidently did for most of the remaining 5 percent — is actually a success. Of the 32,000 people who received the Moderna or Pfizer vaccine in a research trial, only one contracted a severe Covid case.
- To what degree do people who never develop symptoms contribute to transmission? Research early in the pandemic suggested that the rate of asymptomatic infections could be as high as 81%. But a meta-analysis, which included 13 studies involving 21,708 people, calculated the rate of asymptomatic presentation to be 17%.
- The accuracy of rapid testing is questioned – and the more accurate test results are not being given in a timely manner.
- Can children widely spread coronavirus? [current thinking is that they are a minor source of the pandemic spread]
- Why have some places avoided big coronavirus outbreaks – and others hit hard?
- Air conditioning contributes to the pandemic spread.
- It appears that there is increased risk of infection and mortality for those living in larger occupancy households.
- Male patients have almost three times the odds of requiring intensive treatment unit (ITU) admission compared to females.
- Outdoor activities seem to be a lower risk than indoor activities.
Treatments with solid scientific support:
- Dexamethasone
- Proning, or turning someone on their stomach
- Remdesivir
- Baricitinib
Treatments with potential but limited evidence:
- ECMO, or extracorporeal membrane oxygenation
- fluvoxamine
- Cyclosporine
- Famotidine
- Intravenous immunoglobulin
- Ivermectin
- Interferons
Drugs shown to be ineffective:
- The combination of lopinavir-ritonavir
- Hydroxychloroquine
- Insulin
- High dose zinc and vitamin C
- Convalescent plasma
- Monoclonal antibodies
- Tocilizumab
- Anti-coagulants
- A current scientific understanding of the way the coronavirus works can be found [here].
There is now a vaccine available – the questions remain:
- will there be any permanent side effects that will appear months from now,
- how long immunity will last [we can currently say we do not know if it will last more than 4 months],
- there is no solid evidence yet the vaccine will block transmission
Heavy breakouts of coronavirus have hit farmworkers. Farmworkers are essential to the food supply. They cannot shelter at home. Consider:
- they have high rates of respiratory disease [occupational hazard]
- they travel on crowded buses chartered by their employers
- few have health insurance
- they cannot social distance and live two to four to a room – and they eat together
- some reports say half are undocumented
- they are low paid and cannot afford not to work – so they will go to work sick
- they do not have access to sanitation when working
- a coronavirus outbreak among farmworkers can potentially shutter entire farm
The bottom line is that COVID-19 so far has been shown to be much more deadly than the data on the flu. Using CDC data, the flu has a mortality rate between 0.06 % and 0.11 % Vs. the coronavirus which to date has a mortality rate of 4 % [the 4% is the average of overall statistics – however in the last few months it has been hovering around 1.0%] – which makes it between 10 and 80 times more deadly. The reason for ranges:
Because influenza surveillance does not capture all cases of flu that occur in the U.S., CDC provides these estimated ranges to better reflect the larger burden of influenza.
There will be a commission set up after this pandemic ends to find fault [it is easy to find fault when a once-in-a-lifetime event occurs] and to produce recommendations for the next time a pandemic happens. Those that hate President Trump will conclude the virus is his fault.
Resources:
- Get the latest public health information from CDC: https://www.coronavirus.gov .
- Get the latest research from NIH: https://www.nih.gov/coronavirus.
- Find NCBI SARS-CoV-2 literature, sequence, and clinical content: https://www.ncbi.nlm.nih.gov/sars-cov-2/.
- List of studies: https://icite.od.nih.gov/covid19/search/#search:searchId=5ee124ed70bb967c49672dad
include(“/home/aleta/public_html/files/ad_openx.htm”); ?>