Written by Steven Hansen
The U.S. new cases 7-day rolling average is 15.8 % lower than the 7-day rolling average one week ago. U.S. deaths due to coronavirus are now 8.5 % lower than the rolling average one week ago. At the end of this post is a set of interactive graphs and tables for the world and individual states – as well as today’s headlines which include;
- WHO says it’s rare, but possible, that people who have recovered from the coronavirus can get it again
- CDC reverses COVID-19 guidance, says testing may not be needed after exposure
- Drugmaker ramping up production of potential coronavirus vaccine in record time
- Limited legal protection for COVID vaccine makers hampers EU deals
- Alabama research into nasal spray vaccine for coronavirus posts promising results
- Biogen conference may have led to 20,000 COVID-19 infections, researchers say
- Convalescent Plasma: The Unanswered Questions
- Survey finds more than 26,000 coronavirus cases at US universities since pandemic began
- Moderna says vaccine shows promising results in elderly patients

My continuing advice is to continue to wash your hands, wear masks, and maintain social distancing. No handwashing, mask, or social distancing will guarantee you do not get infected – but it sure as hell lowers the risk in all situations. In addition, certain activities are believed to carry higher risk – like being inside in air conditioning and removing your mask (such as restaurants, around your children/grandchildren, bars, and gyms). It is all about viral load.
The daily number of new cases in the U.S. is remaining stubbornly and embarrassedly high.
The following graphs show the 7-day rolling average for new coronavirus cases and deaths have been updated through 26 August 2020:


z coronavirus.png
Coronavirus Statistics For 26 August 2020 |
| U.S. Only | Global | U.S Percentage of Total | ||||
| Today*** | Cumulative | Today*** | Cumulative | Today | Cumulative | |
| New Confirmed Cases | 42,373 | 5,780,000 | 252,711 | 23,930,000 | 16.8% | 24.2% |
| Deaths** | 1,207 | 178,486 | 6,322 | 819,756 | 19.1% | 21.9% |
| Mortality Rate | 2.8% | 3.1% | 2.5% | 3.4% | ||
total COVID-19 Tests per 1,000 people | 1.92* | 220.59* | ||||
* as of 25 Aug 2020
** evidently several states included “probable” deaths today in the number
*** red color indicates record number
Coronavirus News You May Have Missed
- Though rare, it is possible that people who have recovered from Covid-19 could be reinfected, as a recent Hong Kong study suggests, said Maria Van Kerkhove, head of the WHO’s emerging diseases and zoonosis unit.
- Researchers do know that people who are infected develop some antibody response against the disease once they recover. However, researchers don’t yet know how long that immunity lasts, Van Kerkhove said.
- The study also doesn’t change the progress being made toward a coronavirus vaccine, Van Kerkhove added.
CDC reverses COVID-19 guidance, says testing may not be needed after exposure – Yahoo
The Centers for Disease Control and Prevention quietly tweaked its guidance on COVID-19 testing Monday, making a change that could result in fewer people being tested and hinder contact tracing efforts.
The guidance now states that healthy people who have been exposed to COVID-19 “do not necessarily need a test,” as long as they don’t have symptoms. That’s a reversal from previous advice that clearly recommended testing for all close contacts of infected individuals, regardless of whether they had symptoms.
“It seems as though this new guidance is actively discouraging people from seeking testing, even if they have a known exposure,” Angela Rasmussen, a research scientist at Columbia Public Health in New York City, said. “We need more testing, not less.”
… An exposure is considered spending more than 15 minutes in close contact — meaning closer than 6 feet — with a person who has COVID-19. The new recommendation excludes vulnerable individuals, such as older adults and people with chronic medical conditions, who should still be tested after exposure. It also says that state and local health officials may still recommend a test.
Drugmaker ramping up production of potential coronavirus vaccine in record time – CBS
Drugmaker AstraZeneca’s potential coronavirus vaccine is now in advanced trials, and the company says it has the capacity to make 3 billion doses when the vaccine is ready. But even before the final phase of testing and government approval, the vaccine is on a massive manufacturing drive.
… The challenge is how to scale-up from a small vial of vaccine to billions of doses, and quickly.
… That sprint is now a relay race — and a biotech lab on the southern coast of England has the baton. The process starts by making a small batch of vaccine.
“We grow the cells up in this bioreactor and use a starter version of the vaccine, put that in the bioreactor,” Glover said. “It infects the cells that are growing inside here and allows the vaccine to actually make more of the vaccine itself.”
The rest is a complex filtering process that screens out impurities until they are left with a bagful of vaccine ready for vials and, eventually, your arm.
Limited legal protection for COVID vaccine makers hampers EU deals – Reuters
The European Union is offering only partial protection to vaccine makers against legal risks from side-effects of their potential COVID-19 shots, European officials said, in a move that is hampering deals and contrasts with U.S. policy.
With vaccines being developed at record speed during the pandemic, there is potentially a greater risk they may have unexpected consequences or may not be effective. The financial coverage of these liabilities is a key feature of drugmakers’ talks with governments keen to secure vaccine shots in advance.
In these extraordinary circumstances, EU governments “are ready to financially cover certain of the companies’ risks”, an EU official told Reuters. The official added, however, that the EU’s strict rules on liability remain in place.
These rules consider vaccine makers and other manufacturers liable for their products on sale in the EU, apart from rare cases when, for instance, they did not put them into circulation.
The EU’s top court increased the burden on drugmakers in 2017 when it ruled vaccine users were entitled to compensation if they could prove a shot caused a negative side-effect, even when there was no scientific consensus on the matter.
Alabama research into nasal spray vaccine for coronavirus posts promising results – AL
Researchers at the University of Alabama at Birmingham said a nasal spray vaccine candidate for coronavirus showed promising lab results, triggering an immune response in the nose and lungs that could potentially prevent infection.
Scientists have been studying the vaccine in animal models. Studies showed the vaccine stimulated a response by T-cells, which help create antibodies and also attack infected cells the virus uses to create copies of itself.
The T-cells mobilized in the mucus layers of the nose and the lungs, which is where COVID-19 typically takes hold. Troy Randall, an expert on immunology and senior scientist at UAB’s O’Neal Comprehensive Cancer Center, said that sets this particular vaccine apart from those administered as injections.
“With a nasal spray, most of those T-cells will stay in the nose,” Randall said. “And that’s where the virus first lands. So they may be able to attack it and prevent the infection.”
With shots, the body builds a systemic immune response that may take more than a day to reach areas first affected by the virus, Randall said. Lab studies showed a potent response by “killer T-cells” that target infected cells. The vaccine was developed by Altimmune, a company based in Maryland. The company has developed nasal spray vaccines for flu in the past.
“The mucosal T-cell response in the respiratory tract is believed to be dependent on the intranasal route of administration, and we believe it has the potential to provide additional protection against COVID-19,” according to an Altimmune press release. “The induction of a mucosal T-cell response in the lungs has not been shown, to date, with the intramuscularly administered COVID-19 vaccine candidates that are currently in the advanced stages of clinical development.
Biogen conference may have led to 20,000 COVID-19 infections, researchers say – Beckers Hospital Review
A conference held by Cambridge, Mass.-based drugmaker Biogen in a Boston hotel in February may have led to about 20,000 cases of COVID-19 by early May, according to a new study conducted by researchers at the Broad Institute in Cambridge.
The study, which involved more than 50 researchers and that has yet to be peer-reviewed for publication, looked at the genome of the virus in 772 patients in four Massachusetts counties. Of those patients, 289 of them had their infections traced back to the meeting held Feb. 26 and Feb. 27 at the Marriott Long Wharf Hotel in Boston.
By multiplying the proportion of conference-related genomes by the total number of COVID-19 infections in the four counties, the scientists estimated that 20,000 infections could be linked to the Marriott event, which involved about 175 people from the U.S. and overseas.
Of the 289 patients whose virus was traced back to the Biogen event, 122 lived or worked in Boston area homeless shelters, according to The Boston Globe.
The version of the virus found in the 289 patients was one of more than 80 found in the state between late January and early May.
… The researchers emphasized the 20,000 people estimate is a “back-of-the-envelope calculation,” the Globe reported. The actual number could be higher because the researchers didn’t look outside the four counties. It’s also possible the version of the virus they believe stemmed from the conference had spread somewhere else beforehand and then infected people at the Marriott, but the researchers said they saw no evidence to support that.
Convalescent Plasma: The Unanswered Questions – MedPage
Problems with the government’s rationale for authorizing use of convalescent plasma in COVID-19 patients go far beyond the dustup over the purported 35% survival benefit cited by top officials on Sunday, numerous researchers say.
… Much of it came from the Mayo Clinic and FDA expanded access program (EAP), at this point published only as a preprint manuscript. Although a large number of patients were included, the study was observational only, with no untreated control group. That makes the findings merely hypothesis-generating, and can’t offer any firm conclusions. That’s fine for issuing an emergency use authorization (EUA), but not so much for making claims about survival benefit, independent researchers said.
“It’s not even a question of overstating,” Adam Gaffney, MD, MPH, a critical care doctor and health policy researcher at Harvard Medical School, told MedPage Today. “You can’t state much at all when you don’t have a randomized controlled trial.”
“People have made a big deal of Hahn referring to relative versus absolute risk reduction, but I think that’s less of a big deal,” Gaffney said. “The biggest problem is that the data they are citing … is not randomized. That’s the source of all the problems.”
Survey finds more than 26,000 coronavirus cases at US universities since pandemic began – The Hill
A New York Times survey found more than 26,000 coronavirus cases have been reported at U.S. universities since the beginning of the pandemic.
The newspaper surveyed more than 1,500 American colleges and universities to determine that more than 26,000 cases and 64 deaths have been identified at more than 750 institutions.
The data comes as many college students across the nation are returning to in-person instruction and COVID-19 cases have been uncovered in dorms, Greek rows and at college bars in the past few weeks.
Seven large public universities in the South have documented more than 500 cases each, and more than 30 institutions have reported more than 200 each, the Times noted.
The survey included every four-year public institution, every private college that competes in NCAA sports and others that have reported cases.
What is Citriodiol? Insect Repellent Appears to Kill Coronavirus – Newsweek
Researchers in the U.K. have found a Citriodiol, a product found in insect repellant, can kill the coronavirus that causes COVID-19.
Citriodiol is made by Citrefine International Ltd. It is produced from oil from the eucalyptus citriodora tree. The oil is produced by a process that artificially mimics and accelerates the normal acting process in the leaf, converting it into p-menthane-3,8-diol (PMD), according to the company website. This compound is known to have a repellent effect.
The product has approval from the U.S. Environmental Protection Agency.
Previous research had shown Citriodiol can kill SARS CoV-1, the virus that caused the SARS outbreak in 2004. As a result, the U.K.’s Ministry of Defense were looking to see if it had any effect against the new coronavirus.
In April, Sky News announced the armed forces in the U.K. were to be issued with an insect repellent that contains Citriodiol. Because the repellent has no adverse side effects, it was considered a potential way of giving force members an additional layer of protection against COVID-19.
Vaccine Needs 80 Percent Efficiency With 75 Percent Uptake to Stop Pandemic in U.S. – Newsweek
For life to return to “normal,” a coronavirus vaccine will need to have at least 80 percent efficiency, a computer model has found. By simulating different vaccine scenarios in a population, researchers from the U.S. looked at how effective a vaccine would need to be if it were to stop the pandemic alone.
The team, led by Bruce Y. Lee from the CUNY Graduate School of Public Health and Health Policy, found that if 75 percent of the population got the vaccine, 80 percent effectiveness would stop an ongoing epidemic. If only 60 percent got the vaccine, it would need to have 100 percent efficiency to extinguish the epidemic. Their findings are published in the American Journal of Preventive Medicine.
The computer model simulated the spread of COVID-19 and vaccination across the U.S. At present, there are no approved treatments for the disease, and current control methods are largely reliant on non-pharmaceutical interventions, such as social distancing, lockdowns and mask use. Because of this, there has been a huge focus on the development of a vaccine.
Moderna says vaccine shows promising results in elderly patients – CNBC
Moderna’s potential Covid-19 vaccine generated a promising immune response in an early-stage trial of elderly patients, according to new preliminary data.
The company tested its vaccine on 10 adults between the ages of 56 and 70, and 10 elderly adults aged 71 and older. Each participant received two 100 microgram doses of the vaccine 28 days apart.
The volunteers produced neutralizing antibodies, which researchers believe are necessary to build immunity to the virus, and killer T-cells, Moderna said. Additionally, the antibodies that were produced were higher than those seen in people who have recovered from Covid-19. The vaccine also appeared to be well tolerated. Some patients reported fatigue, chills, headaches and pain at the injection site, though the majority of symptoms resolved within two days, the company said.
The following are foreign headlines with hyperlinks to the posts
Spain ready to send in troops to trace resurging coronavirus
Sweden Finds Thousands of False Positives From Chinese Virus Test Kits
Beijing Now Coronavirus-Free as Last Two Patients Discharged From Hospital
In Germany, early results of school reopenings are hopeful, but it’s ‘messy and imperfect.’
Kenya’s president extends a nationwide 9 p.m. curfew for another 30 days.
India approves first nationally made Covid-19 test
India has recorded over 1.5 million new Covid-19 cases in August alone
The following are additional national and state headlines with hyperlinks to the posts
6 days after reopening, the University of Alabama has over 500 coronavirus cases.
Flu Season Could Make Coronavirus Testing Delays Even Worse
U.S. rejects U.N. rights panel upholding access to abortions during pandemic
More Than 200 Ohio State University Students Suspended For Violating Pandemic Rules
MTA warns of 40 percent reduction in New York subway and bus service, thousands of job losses
Oahu restores stay-at-home-order
Florida confirmed 9K new COVID-19 cases among children within 15 days as schools reopen
Bed Bath & Beyond is laying off 2,800 employees
Today’s Posts On Econintersect Showing Impact Of The Pandemic With Hyperlinks
July 2020 Coincident Indices Mostly Remain Infected By Coronavirus
Headline Durable Goods New Orders Improved Again In July 2020
The Effects Of Extra Unemployment Benefits On Household Delinquencies
Debt Relief And The CARES Act: Which Borrowers Benefit The Most?
July 2020 Headline New Home Sales Get Stronger
Coronavirus INTERACTIVE Charts
include($_SERVER[‘DOCUMENT_ROOT’].’/pages/coronavirus.htm’); ?>
Analyst Opinion of Coronavirus Data
There are several takeaways that need to be understood when viewing coronavirus statistical data:
- The global counts are suspect for a variety of reasons including political. Even the U.S. count has issues as it is possible that as much as half the population has had coronavirus and was asymptomatic. It would be a far better metric using a random sampling of the population weekly. In short, we do not understand the size of the error in the tracking numbers.
- Just because some of the methodology used in aggregating the data in the U.S. is flawed – as long as the flaw is uniformly applied – you establish a baseline. This is why it is dangerous to compare two countries as they likely use different methodologies to determine who has (and who died) from coronavirus.
- COVID-19 and the flu are different but can have similar symptoms. For sure, COVID-19 so far is much more deadly than the flu. [click here to compare symptoms]
- From an industrial engineering point of view, one can argue that it is best to flatten the curve only to the point that the health care system is barely able to cope. This solution only works if-and-only-if one can catch this coronavirus once and develops immunity. In the case of COVID-19, herd immunity may need to be in the 80% to 85% range. WHO warns that few have developed antibodies to COVID-19. At this point, herd immunity does not look like an option although there is now a discussion of whether T-Cells play a part in immunity [which means one might have immunity without antibodies]
- Older population countries will have a higher death rate.
- There are at least 8 strains of the coronavirus. New York may have a deadlier strain imported from Europe, compared to less deadly viruses elsewhere in the United States.
- Each publication uses different cutoff times for its coronavirus statistics. Our data uses 11:00 am London time. Also, there is an unexplained variation in the total numbers both globally and in the U.S.
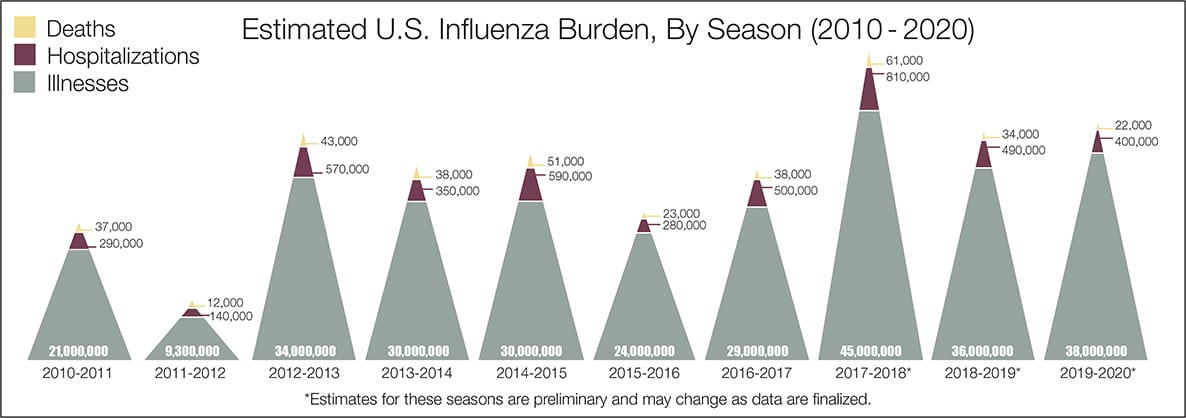
- The real question remains if the U.S. is over-reacting to this virus. The following graphic from the CDC puts the annual flu burden in perspective [click on image to enlarge]. Note that using this data is dangerous as the actual flu cases are estimated and not counted – nobody knows how accurate these guesses are.
What we do not know about the coronavirus [actually there is little scientifically proven information]. Most of our knowledge is anecdotal, from studies with limited subjects, or from studies without peer review.
- How many people have been infected as many do not show symptoms?
- Masks do work.
- Do we develop lasting immunity to the coronavirus? Another coronavirus – the simple cold – does not develop long term immunity.
- To what degree do people who never develop symptoms contribute to transmission?
- The US has scaled up coronavirus testing – and the accuracy of the tests has been improving. However, if one loses immunity – the coronavirus testing value is reduced.
- Can children widely spread coronavirus? [current thinking is that they are becoming a major source of the pandemic spread]
- Why have some places avoided big coronavirus outbreaks – and others hit hard?
- What effect will the weather have? At this point, it does not seem hot weather slows this coronavirus down – and it seems air conditioning contributes to its spread.
- Outdoor activities seem to be a lower risk than indoor activities.
- Can the world really push out an effective vaccine in 12 to 18 months?
- Will other medical treatments for Covid-19 ease symptoms and reduce deaths? So far only one drug (remdesivir) is approved for treatment.
- A current scientific understanding of the way the coronavirus works can be found [here].
Heavy breakouts of coronavirus have hit farm workers. Farmworkers are essential to the food supply. They cannot shelter at home. Consider:
- they have high rates of the respiratory disease [occupational hazard]
- they travel on crowded buses chartered by their employers
- few have health insurance
- they cannot social distance and live two to four to a room – and they eat together
- some reports say half are undocumented
- they are low paid and cannot afford not to work – so they will go to work sick
- they do not have access to sanitation when working
- a coronavirus outbreak among farmworkers can potentially shutter entire farm
The bottom line is that COVID-19 so far has been shown to be much more deadly than the data on the flu. Using CDC data, the flu has a mortality rate between 0.06 % and 0.11 % Vs. the coronavirus which to date has a mortality rate of 4 % [the 4% is the average of overall statistics – however in the last few months it has been hovering around 1.0%] – which makes it between 10 and 80 times more deadly. The reason for ranges:
Because influenza surveillance does not capture all cases of flu that occur in the U.S., CDC provides these estimated ranges to better reflect the larger burden of influenza.
There will be a commission set up after this pandemic ends to find fault [it is easy to find fault when a once-in-a-lifetime event occurs] and to produce recommendations for the next time a pandemic happens. Those that hate President Trump will conclude the virus is his fault.
Resources:
- Get the latest public health information from CDC: https://www.coronavirus.gov .
- Get the latest research from NIH: https://www.nih.gov/coronavirus.
- Find NCBI SARS-CoV-2 literature, sequence, and clinical content: https://www.ncbi.nlm.nih.gov/sars-cov-2/.
- List of studies: https://icite.od.nih.gov/covid19/search/#search:searchId=5ee124ed70bb967c49672dad
include(“/home/aleta/public_html/files/ad_openx.htm”); ?>